|
In such cases the need for medical intervention is obvious. Some patients do not present until they have developed widespread peripheral oedema. Overt cardiogenic peripheral oedema develops because the fluid retention results in an increase in intravascular hydrostatic pressure and a commensurate increase in the filtration rate, which eventually exceeds the capacity of the lymphatics to drain fluid away (see Figure 1). Any filtered fluid is then drained by the lymphatics. In normal circulation, there is continuous filtration of fluid from the intravascular space into the tissues at a rate dependent on the gradient between the intravascular and extravascular hydrostatic pressure. The accumulation of fluids is a gradual process. Glomerular filtration rate falls, enhancing and perpetuating the vicious cycle. The elevated VP can further reduce renal blood flow as the gradient between mean renal arterial pressure (often itself decreased by the HF process) and VP declines. The heart itself tends to worsen with time as the failing LV tends to dilate, as does the left atrium, particularly if mitral regurgitation develops. The activation of the renin–angiotensin–aldosterone and AVP systems maintain cardiac preload (more fluids) and afterload (vasoconstriction, mainly due to angiotensin II), thereby maintaining the homeostasis of the cardiovascular system but at a cost of increased systemic venous pressure (VP). 7 Arginine vasopressin (AVP) is also released, 8,9 further enhancing fluid retention and stimulating thirst. The kidneys respond by increasing the production of renin, leading to more aldosterone production, which is consequently followed by sodium and water retention. As the heart starts to fail, renal perfusion falls. The development of peripheral oedema in patients with HF is related to fluid excess. 
Why Do Patients with Heart Failure Retain Fluid? However, for many patients, some degree of congestion remains even with treatment, 5,6 and it is not clear how many patients with CHF have subclinical congestion – that is, have an excess of body fluid falling short of the volume required to cause overt peripheral oedema. The aim of management is to remove the excess fluid, so that the patient is no longer congested when they leave hospital, now transitioning to a diagnosis of ‘chronic HF (CHF)’. 4 Peripheral congestion in patients with heart failure usually develops over weeks or even months, and patients may present ‘acutely’ having gained over 20 litres of excess fluid, and hence over 20 kg of excess weight. 3 For most patients, however, congestion is a more generalised process that usually develops more gradually (peripheral oedema), and its management will be the focus of discussion in this review.Ĭhronic fluid accumulation is responsible for a substantial number of hospital admissions, and identifies patients with a worse prognosis than those admitted due to a sudden increase in LV filling pressures. In this circumstance, the oedema is localised predominantly to the pulmonary airspaces (pulmonary oedema), while the total amount of fluid in the cardiovascular system remains unchanged. In some patients, pulmonary congestion develops very rapidly because of a sudden increase in LV filling pressures, and a precipitating factor is often recognised, such as acute myocardial ischaemia, or uncontrolled hypertension. 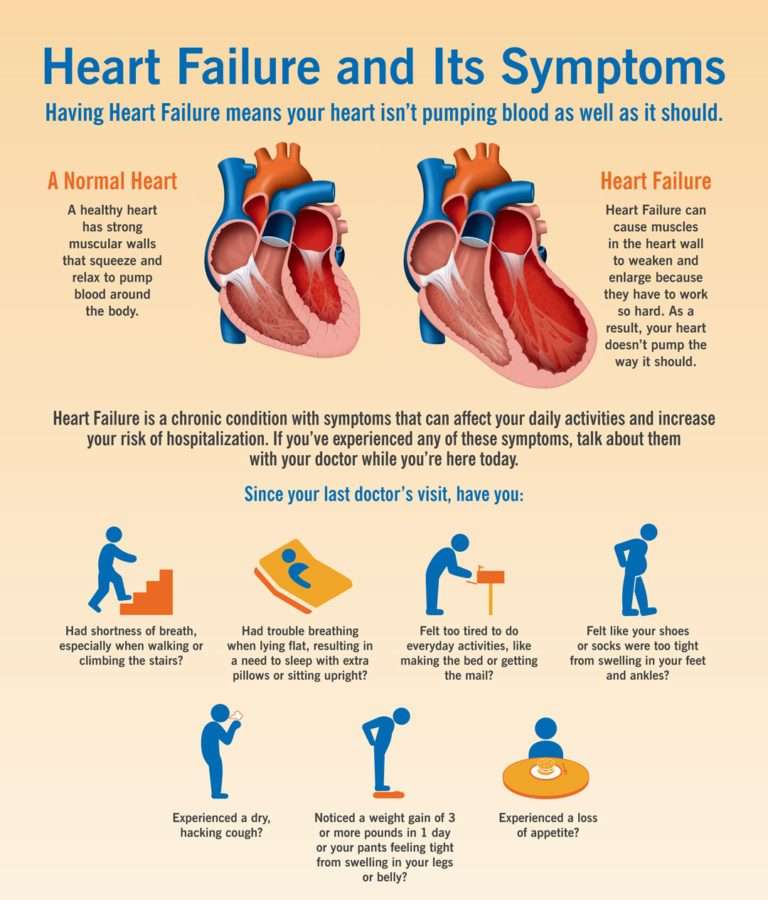
1,2 Congestion, or fluid overload, is a classic clinical feature of patients presenting with HF. It is associated with long in-patient stays, and has a high in-hospital and post-discharge morbidity and mortality, whether left ventricular ejection fraction (LVEF) is reduced (HFREF) or normal (HeFNEF). Heart failure (HF) is one of the most common reasons for admission to hospital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
